|
11/6/2022 0 Comments Atomic orbitals Approximate expansion of the molecular orbitals over a much reduced set of orthogonal atomic basis functions can also be accomplished to a very good accuracy with a singular value decomposition procedure. bonds occur when orbitals overlap between the nuclei of two atoms, also known as the. Both bonds are formed from the overlap of two orbitals, one on each atom. For example, the orbitals with (l0) are called s orbitals those with (l1), p orbitals those with (L2), d orbitals, etc. Recall that each value of l is assigned a letter that corresponds to particular orbitals. There are two types of overlapping orbitals: sigma ( ) and pi ( ). The shape of an atomic orbital is determined primarily by l, the angular momentum quantum number. In addition, the molecular orbitals of a calculation can be exactly expressed as a linear combination of this orthonormalized set of numerical atomic orbitals, and the Mulliken population analysis carried out on this basis set exactly reproduces the original QTAIM atomic populations of the atoms. This partial merging of atomic orbitals is known as orbital overlapping or overlapping of atomic orbitals. The occupation numbers of the remaining effective orbitals are almost negligible, except for atoms with hypervalent character. This tutorial examines the first four energy levels of an atom, s, p, d, and f, chosen through the pull-down menu. These correspond to atomic hybrids that closely resemble the core and valence shells of the atom. Experience shows that only a limited number of effective atomic orbitals exhibit significant occupation numbers. This formalism can be used to retrieve from any type of calculation a proper set of orthonormalized numerical atomic orbitals, with occupation numbers that sum up to the respective Quantum Theory of Atoms in Molecules (QTAIM) atomic populations. 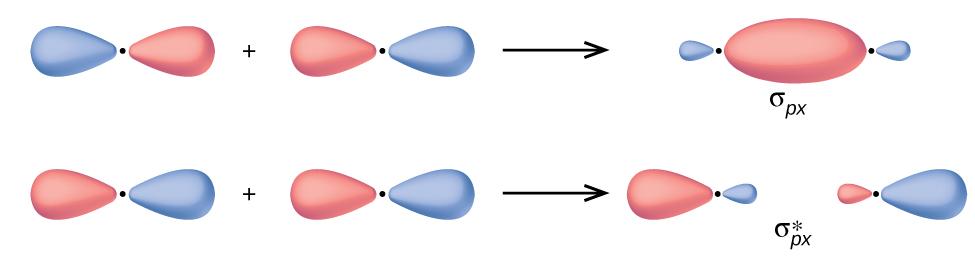 
The effective atomic orbitals have been realized in the framework of Bader's atoms in molecules theory for a general wavefunction. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
